Tracheal Mites (The Isle of Wight Disease That Stumped Beekeepers for Decades)
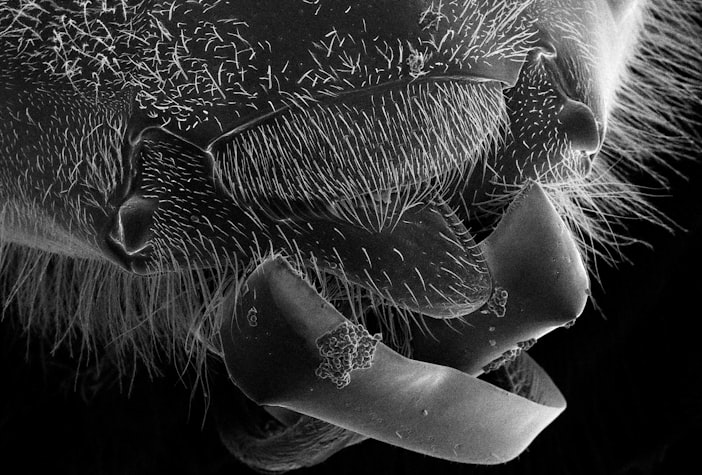
The mite lives inside the bee's breathing tubes. Not on the bee - inside the actual airways. Acarapis woodi enters through a spiracle (one of the external breathing holes on the bee's thorax), punctures the tracheal wall with its mouthparts, feeds on bee blood, and lays eggs in the airway. Its offspring hatch inside the same airway. The whole life cycle - birth, development, mating, reproduction - happens inside another animal's respiratory tract.
The mature female is approximately 120 to 175 micrometers long. About the width of two human hairs. You cannot see her without a microscope. She can, over the course of several generations inside a single bee, clog and discolor the tracheal tubes until the bee's flight muscles are oxygen-deprived and the bee can no longer fly.
When enough bees in a colony are carrying this - particularly the long-lived winter bees who can't be replaced easily in cold months - the colony loses its capacity to generate the metabolic heat that keeps it alive through winter. The cluster cools. Everything dies.
This happened to 90 percent of British and Irish honey bee colonies between 1906 and the early 1920s, and nobody knew why for 15 years.
The Isle of Wight Mystery
In 1906, beekeepers on the Isle of Wight noticed bees crawling on the ground in front of their hives, unable to fly. Wings were held at odd angles - what would later be called K-wing, because of the shape - rather than folded neatly over the abdomen. Colonies weakened and died. The die-off spread from the island to the mainland and across the British Isles.
Theories ranged from a new virus to environmental toxins to genetic degeneration. The epidemic was called "Isle of Wight disease," and it was the Colony Collapse Disorder of its era - mass die-off, no identified cause, more theories than evidence. An estimated 90 percent of British and Irish colonies were lost over the following decade and a half.
In 2026, John Rennie, a zoologist at the University of Aberdeen, dissected affected bees and found them: microscopic mites living inside the prothoracic tracheae - the first and largest pair of breathing tubes in the bee's thorax. He named the mite Acarapis woodi, honoring his colleague E.A.N. Wood. The Isle of Wight disease had a cause, and it was invisible to the naked eye.
The Apiary Project tracks every major threat to US honey bee colonies.
Explore the Issues Hub →How It Works
The life cycle unfolds entirely inside the host. A mated female mite enters a young bee's spiracle within the first four days of the bee's adult life - older bees develop stiffer tracheal hairs that block entry. Once inside, she punctures the tracheal wall, begins feeding, and lays five to seven eggs over her lifetime. The eggs hatch and develop inside the same trachea. When the next generation matures and mates, the young females exit through the spiracle, crawl across the bee's exterior, and transfer to a new young bee through direct physical contact - the kind that happens constantly during in-hive activities like nursing, food exchange, and grooming.
A heavily infested bee suffers from three overlapping problems. The mites, their eggs, their feces, and the hemolymph leaking from punctured tracheal walls accumulate inside the airway. Healthy tracheae are clear or pale amber with visible spiral reinforcements; infested ones are brown to black, visibly fouled under a dissecting microscope. The obstruction reduces airflow. The feeding punctures drain hemolymph - the insect's blood, which carries nutrients, hormones, and immune cells. And because insects don't use blood to transport oxygen (oxygen diffuses directly through the tracheal tubes to the cells), blocked tubes mean oxygen-starved flight muscles, which produce K-wing: the bee crawls rather than flies.
Tracheal mite kills tend to happen in late winter, when the mite population has been building inside long-lived winter bees for months and the colony's demand for thermoregulatory heat is highest. The cumulative damage reaches a threshold at precisely the moment when the colony is most vulnerable.
The American Arrival
Acarapis woodi was first detected in the United States in 1984, in colonies in Weslaco, Texas. The mite had been in Mexico since at least the 1960s and entered the US through migratory beekeeping or natural movement across the border.
The US had been the last major beekeeping country without tracheal mites. The Honeybee Act of 1922 - a federal import ban designed to keep foreign bee parasites and diseases out - had held the mite at bay for 60 years. It would fail for Varroa, which arrived through other channels in the mid-1980s, but it worked for tracheal mites for six decades. That counts as a success, in the context of what usually happens when parasites are moving.
The initial US impact was severe. Colony losses spiked. Winter mortality climbed. Beekeepers in cold northern states - where winter bee longevity is critical - were hit hardest. In the late 1980s, the tracheal mite crisis was described in the same alarmed terms that would later apply to Varroa and CCD.
Two Treatments, One Memorable
Two treatments became standard. The first is menthol: crystalline menthol placed on the hive's top bars sublimates into vapor that fills the hive interior, enters the bees through their spiracles, and kills or repels the mites inside. It works. It's derived from mint. It doesn't contaminate honey if supers aren't present. The EPA approved it in 2026.
The second is grease patties. Vegetable shortening mixed with granulated sugar, formed into patties, placed on the top bars. The grease coats the bees as they contact it, and when young mated females exit one bee's trachea and walk across the exterior to find a new host, they slip on the greasy surface and can't grip. The transfer fails. The mite population growth slows.
A beekeeper asked to explain how they treat a microscopic parasite living inside another animal's breathing tubes sometimes pauses before saying "vegetable shortening." The answer is accurate. Beekeeping is full of solutions that work despite sounding made up.
Neither treatment eliminates tracheal mites entirely. Both reduce infestation to manageable levels. The approach is suppression, not eradication - a recurring theme in bee parasite management.
Brother Adam and the Buckfast Bee
Brother Adam - Karl Kehrle, a Benedictine monk at Buckfast Abbey in Devon, England - watched the Isle of Wight disease destroy British beekeeping in the early 20th century and spent the rest of his life breeding a bee that could survive it. His program, which produced the Buckfast bee, ran for over 70 years. He traveled more than 100,000 miles across Europe, North Africa, and the Middle East collecting bee strains, selecting for tracheal mite resistance, gentleness, and productivity.
The Buckfast bee demonstrates significant resistance to tracheal mite infestation. The mechanism appears behavioral: Buckfast bees groom more actively, removing mites from their bodies before they can enter the spiracles. The trait is heritable and has been maintained in Buckfast lines across decades of breeding.
In the US, the USDA identified tracheal mite resistance in Russian bee stocks from the Primorsky region, where the bees had presumably coexisted with Acarapis mites or close relatives long enough to develop behavioral defenses. Coevolution produces resistance, given enough time. The problem is that the parasite usually arrives before the coevolution can happen.
The Forgotten Mite
Then Varroa arrived, and tracheal mites became yesterday's problem.
Varroa was bigger - visible without a microscope. Varroa was faster - killing colonies more reliably and at higher rates. Varroa vectored viruses that produced dramatic symptoms. Varroa rewrote beekeeping economics and demanded entirely new management strategies. Every research grant, every extension publication, every conversation at the beekeeping club shifted to Varroa. Tracheal mites became a footnote.
They're still present in US colonies. Still infesting, still feeding, still clogging tracheae. But the relative impact of tracheal mites next to Varroa is like comparing a dripping faucet to a burst pipe. There's some evidence their prevalence declined as Varroa treatments (Apistan, CheckMite+) had incidental effects on them as well. Breeding programs selecting for Varroa resistance may have inadvertently selected for grooming behavior that also reduces tracheal mite infestation. The fight against the bigger mite may have accidentally helped with the smaller one.
Diagnosing tracheal mites still requires dissection: collect 30 to 50 bees, freeze them, cut through the collar, expose the prothoracic tracheal trunks under a dissecting microscope, and check for discoloration. Clear or pale amber is healthy. Brown to black is infested. It's slow, exacting work that requires practice and equipment, in an era when most beekeeping attention is elsewhere.
The tracheal mite is still there. Still the width of two human hairs, still piercing tracheal walls, still completing its life cycle inside another animal's breathing tubes, in bees that are already fighting a harder fight. The first mite crisis taught the beekeeping world what a new parasite can do. The second mite crisis arrived before the lesson had fully settled.
The annual colony loss data tracked by USDA and the Bee Informed Partnership doesn't distinguish tracheal mite mortality from Varroa mortality in most analyses - the latter is so dominant that the former gets absorbed into the background noise. The Apiary Project tracks colony health data as part of monitoring which pressures are currently driving losses in US populations.
The tracheal mite crisis created the Buckfast bee, the menthol treatment, and the grease patty. Then a larger crisis arrived, and the smaller one became the one you mention in passing, after the main discussion is already over.